
“Breakthroughs that, not long ago, existed only in theory or distant ambition are rapidly becoming a reality.”
Dr. Mel Spigelman,
President & CEO, TB Alliance
Tuberculosis remains one of the world’s gravest health and economic threats, but innovative science is powering solutions.
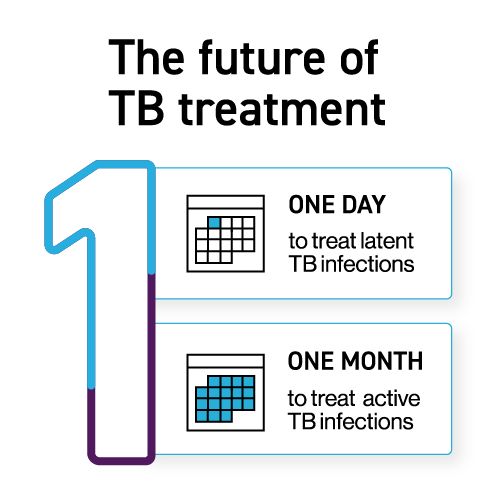
Every year, tuberculosis (TB) continues to claim more lives than any other infectious disease worldwide. Despite decades of progress, TB remains a critical threat to global health, and many still face barriers to timely diagnosis and treatment. But through science and partnership, that burden is beginning to shift – especially for drug-resistant TB. TB Alliance is working to revolutionize TB treatment outcomes and experiences: on a path to one-day treatment for latent TB, one-month treatment for active TB, and ensuring access for all, even those often left behind in research and delivery systems.
A new generation of faster, simpler TB cures is taking shape today.
2025 Highlights
Science:
Unlocking Faster Cures
In 2025, TB Alliance advanced its 1×1 mission with a successful Phase 2 clinical trial of the SPaL regimen (a combination of sorfequiline, pretomanid, and linezolid), which outperformed standard treatment after just two months. These results set the stage for a pivotal Phase 3 clinical trial, with additional progress toward applying long-acting injectable technology to next-generation cures, the path to a one-month TB cure becomes clearer and more achievable.
Impact:
A Blueprint for Global Access
In 2025, pretomanid’s reach accounted for approximately 80% of the drug-resistant cases treated annually. New initiatives like the PeerLINC Knowledge Hub and tools like Upskill TB helped frontline providers and health systems expand access to the BPaL/M regimens, saving more than 20,000 lives and $160 million globally, while demonstrating how thoughtful access planning accelerates and amplifies impact.
Community:
Voices Leading the Way
In 2025, communities around the world continued to play a powerful role in advancing the TB response. From clinical trial participants and community engagement leaders to donors, advocates, and activists, their efforts helped raise awareness, build trust, and drive support and demand for faster, more people-friendly TB cures. Public voices like author John Green brought new audiences into the conversation, while longstanding champions ensured the momentum continued across countries and sectors.

“The speed and scale of what TB Alliance has achieved is only possible because of a shared commitment – to bold innovation, to collaboration, and to empowering TB affected communities to help realize the future of TB treatment.”
David Norton
Chair, TB Alliance Board of Directors
Driving Change Through Collaboration, Commitment, and Courage
But the work is far from done. With continued investment, scientific ambition, and community-driven solutions, we can deliver on the promise of truly transformative TB cures – for everyone, everywhere.
SCIENCE
Unlocking the Future of TB Treatment
Results from NC:009 Show Potential to Shorten TB Treatment Time
Results from a Phase 2 clinical trial provided the most promising evidence yet for the potential of a new regimen featuring sorfequiline, a novel diarylquinoline being developed by TB Alliance. When combined with pretomanid and linezolid (in a regimen called “SPaL”), the three drugs demonstrated higher efficacy than the current standard of care after two months of treatment for drug-sensitive TB. The trial also confirmed a comparable safety and tolerability profile. These findings mark a critical step toward significantly shorter TB therapies. Based on these results, our researchers are preparing for a Phase 3 trial to further evaluate the next step in developing this treatment.
Promising New Data Presented on Pediatric Use of Pretomanid
At the 2025 Union Conference, TB Alliance shared encouraging new data on the use of pretomanid in children with drug-resistant tuberculosis. Interim results from a Phase 1 trial showed that pediatric formulations of pretomanid were well tolerated, with drug exposure levels aligning with those seen in adults. The study is part of ongoing efforts to develop child-friendly, all-oral treatment options for TB. These findings underscore the potential of pretomanid to become a key component in shorter, more manageable regimens for children – supporting TB Alliance’s mission to expand access to innovation across all age groups.

New Data Reveals Key TB Alliance Drugs and Drug Candidates are Well-Suited for Development as Long-Acting Injectables
At the 2025 Union Conference, TB Alliance presented promising preclinical data on long-acting injectable (LAI) formulations for TB. These therapies, designed to maintain therapeutic drug levels for months from a single injection, could revolutionize treatment for both active and latent TB, bringing within reach one-shot cures TB cures that can simplify care and improve adherence. Lead candidates—including sorfequiline, pretomanid, and TBAJ-587—demonstrated sustained systemic exposure for up to four months in animal models.


Developing New Treatment Options Beyond TB
TB Alliance is applying its scientific expertise beyond TB, targeting related diseases like Buruli ulcer, leprosy, and nontuberculous mycobacterial infections (NTMs) – all caused by bacteria with similar biological blueprints to TB. With limited treatments and funding available for these neglected diseases, TB Alliance is exploring whether TB drug candidates can be leveraged to help. A Phase 2 trial is already underway testing telacebec for Buruli ulcer, with the potential to cut treatment time in half. This expansion not only brings hope to overlooked patient populations, it may also reveal new insights that feed back into the fight against TB itself.

SPOTLIGHT: Thuto Pulane said, “Don’t Panic; TB is not the end of the road”
Thuto Pulane, a TB survivor and participant in the NC-009 clinical trial, received treatment under Dr. William Brumskine at the Aurum Institute Clinical Research Site in Rustenberg, South Africa. Her experience reflects the vital role of clinical trial participants in advancing research – and the hope that faster, simpler cures can offer to people affected by TB.

“The treatment was amazing. It was shorter and easier than I expected. I’d tell anyone not to be afraid. TB treatment has come a long way, and this new research gives us even more hope.”
Thuto Pulane
TB survivor and participant in the NC-009 clinical trial
IMPACT
The Growing Impact of BPaL/M: 20,000 Lives and $160M saved and counting…
Through 2025, 246,000 courses of pretomanid had been procured across more than 110 making pretomanid roll-out the fastest for a new TB medicine in modern times. In collaboration with TB Alliance, Johns Hopkins University conducted a modeling exercise using published data and projections to quantify the impact of the BPaL/M regimens to-date and projected over the next 10 years. Per this model, BPaL/M has already saved 20,000 lives and about $160 million compared to the continued use of alternate treatments.
In 2025, large and highest burden countries across the world began rolling out BPaL/M. India, Brazil, and China held a series of BPaL/M trainings among their respective experts group, setting the stage for future scale up. Several other high-burden countries e.g., Indonesia, Pakistan, Peru, Philippines, South Africa, and Ukraine began offering BPaL/M as the default treatment to all new and eligible patients.
In lockstep with increasing usage, availability and affordability of pretomanid continued to improve – a third TB Alliance licensee, Lupin Labs, entered the market by gaining WHO pre-qualification and prices dropped further by 33% to under US$1/tablet (88c/tablet) due to generic competition.
TB Alliance worked with partners on multiple initiatives to support rapid scale-up on novel TB treatments including BPaL/M and other regimens for special, niche populations as recommended by WHO.
We cannot afford to stop until our treatments are available to everyone who needs them. To date, new, six-month treatments for DR-TB have been procured to reach
246K
patients
By 2034 BPaL/M will…

PDPs as Orchestrators of Access: BPaL/M is a Blueprint for Fast, Equitable Access to TB Innovation
In 2025, the Geneva Graduate Institute developed a case study and analysis that highlighted how TB Alliance was able to achieve unprecedented speed and scale in expanding access to pretomanid and the BPaL/M regimens – reaching more than 100 countries in the shortest ever timeframe for a TB treatment. The study found that TB Alliance acted as a highly effective orchestrator reaching across the access landscape to align regulatory, market, and country-level efforts, while convening national TB programs, technical partners, stakeholders, , global policy makers, manufacturers, and communities to drive rapid uptake. Crucially, the report makes a strong case for donors to invest not only in R&D, but also in the robust access programs of product development partnerships – where well-resourced access work can dramatically accelerate impact and save lives.

700+
experts
Faster, Fairer, More Affordable Technical Assistance
Launched in Manila, Philippines in 2024, TB Alliance’s PeerLINC Knowledge Hub helps countries accelerate adoption of new TB technologies by connecting them with “early adopter” peers for practical, country-driven training. Compared with traditional technical assistance, PeerLINC’s peer-based approach is more responsive, cost-effective, and equitable for countries of different sizes, combining classroom learning with on-the-ground, context-specific support through partnerships in the Philippines and alumni countries. Through 2025, PeerLINC had:
- Trained more than 700 experts in 41 countries.
- Built G-TEC, a network of 40 global experts from 17 countries to help clinicians worldwide to with managing complex DR-TB cases.
- Expanded training offerings to include technologies beyond BPaL/M.
- Expanded the pool trainers involving experts from alumni countries.
Scalable, Tech Enabled Platforms Help Power the TB Treatment Revolution
Historically, breakthrough TB innovations take too long to reach those who need them. With the introduction of pretomanid and the BPaL/M regimens, TB Alliance changed that. A major driver of this accelerated rollout is a suite of scalable, cost-efficient tech-enabled health tools focused on enabling frontline health providers, and co-developed with high-burden countries. These tools are supporting rapid adoption and scale-up of BPaL/M and can also be adapted for other TB treatments.
- Upskill TB is a digital learning platform to strengthen healthcare capacity to treat DR-TB with modern tools
- Manage TB is an e-prescription platform that serves as a clinical decision support for clinicians treating DR-TB in India
- SLASH-TB is an economic modeling tool that has been used by multiple governments and health programs to quantify the value proposition of implementing new DR-TB treatments thereby supporting budget and prioritization decisions for BPaL/M. It has been expanded to model new pan-TB LAI regimens.
- Fast Track the Cure is a community engagement initiative that has mobilized communities in nine high-burden countries. It has helped accelerate access to improved DR-TB treatments by advancing treatment literacy, generating demand for new treatments, and capacitating community-led monitoring.




WHO Recognizes Successful Rollout of BPaL/M as Critical in Fight Against AMR
Antimicrobial resistance (AMR) is a growing global health threat, and drug-resistant TB is its deadliest form. In 2025, the World Health Organization’s Global AMR Report highlighted TB Alliance’s work to expand access to the BPaL/M regimens as an example of how innovation combined with deliberate access planning can accelerate the responsible introduction of new antibiotics.
The report acknowledges TB Alliance’s leadership in executing a coordinated strategy that engaged high-, moderate-, and lower-burden countries within a short three- to five-year period, TB Alliance helped transform the market for pretomanid from a small and uncertain one into a sustainable and viable global treatment option. By working closely with WHO and national stakeholders, supporting early policy and regulatory adoption, building programmatic capacity, and shaping the market for responsible scale-up, TB Alliance helped ensure that this critical regimen reaches patients who need it most. Today, BPaL/M is being used in more than 100 countries and is delivering cure rates above 90%, demonstrating how coordinated innovation and access efforts can help address the growing challenge of AMR.
SPOTLIGHT: 10 Years Ago They Faced DR-TB, Today They Thrive: Catching Up with Participants from the Trial that Led to Approval of BPaL
The Nix-TB trial was a turning point in the fight against drug-resistant tuberculosis; it was the Phase 3 trial that collected the data upon which pretomanid and BPaL earned initial regulatory approval. That trial was initiated in 2015. A decade later, we revisited three of the trial’s participants to see how their lives have changed since completing treatment. To see people who were once facing a terrifying diagnosis and lengthy, arduous, and unreliable treatment now thriving, growing their families, and building their businesses is a reminder of the power of new TB cures’ ability not just to save, but to transform lives.
COMMUNITIES
Progress Powered by Partnership
These advances were not achieved alone. They reflect the extraordinary commitment of our research and access partners, the sustained support of our funding partners, and—above all—the courage and contributions of clinical trial participants. Together, even in challenging times, we continue to push further to spearhead new and improved cures and translate scientific innovation into real-world impact.






Communities in the Lead: Fast Track the Cure Welcomes Six New Partners
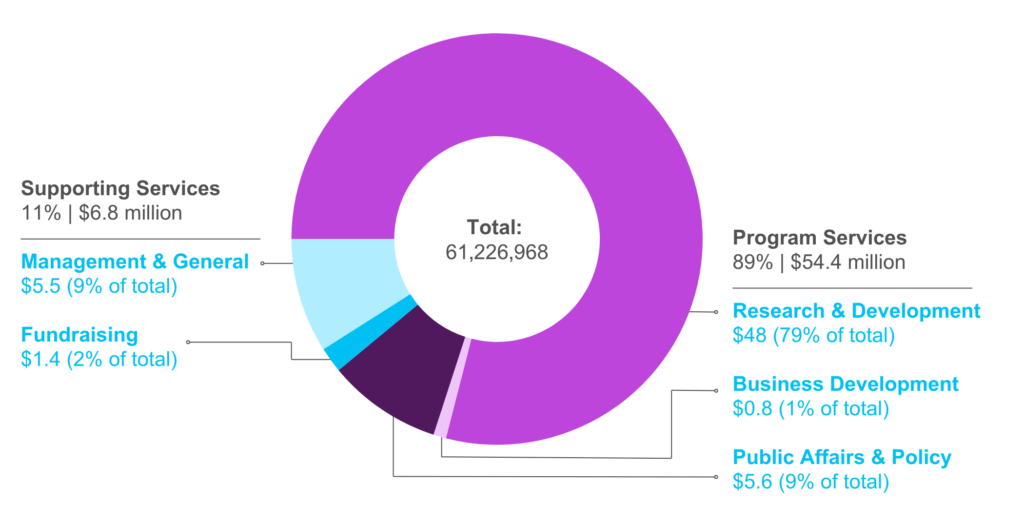
Our Financials
Much has been made of the retraction of global funding from the United States, but this shift is part of a broader trend that predates 2025. TB Alliance continues to pursue innovative partnerships that provide mutual value for investment, including increased engagement of high-burden TB countries in research and access collaborations. While funding priorities and trends may evolve over time, as outlined in a 2025 Impact Global Health report, several truths remain: investing in global health R&D delivers extraordinary economic returns for both traditional donor countries and endemic countries; innovations developed for neglected diseases often generate “boomerang” health and economic benefits in donor countries; and global health R&D is a pillar of national security and pandemic preparedness.




New Partnerships for Innovative Approaches
TB Alliance also worked with manufacturing partners to streamline procurement of pretomanid in Canada. Additional partnerships included Afrigen Biologics and Vaccines and the Desmond Tutu TB Centre (DTTC) at Stellenbosch University joining TB Alliance’s Global TB Stakeholders.
SPOTLIGHT: Dr. William Brumskine Talks about the Importance of Communities
Dr. William Brumskine, principal investigator of the NC-009 trial at the Aurum Institute Clinical Research Site in Rustenburg, South Africa, chatted with us about the vital role of community engagement in TB research. From consulting community advisory boards to ensuring clear communication with participants, he explains how meaningful partnerships with local communities help shape ethical, effective clinical trials and ensure that research ultimately benefits the people it is meant to serve.