Leveraging Resources
Funding new treatments
TB Alliance makes every effort to ensure that donor money is put to effective use and contributes directly to the development of impactful new TB drug regimens that are adopted, available and affordable.
TB Alliance Spending

Stepping up: World Leaders Express Commitment to Fund TB Research; Accountability Will Be Key
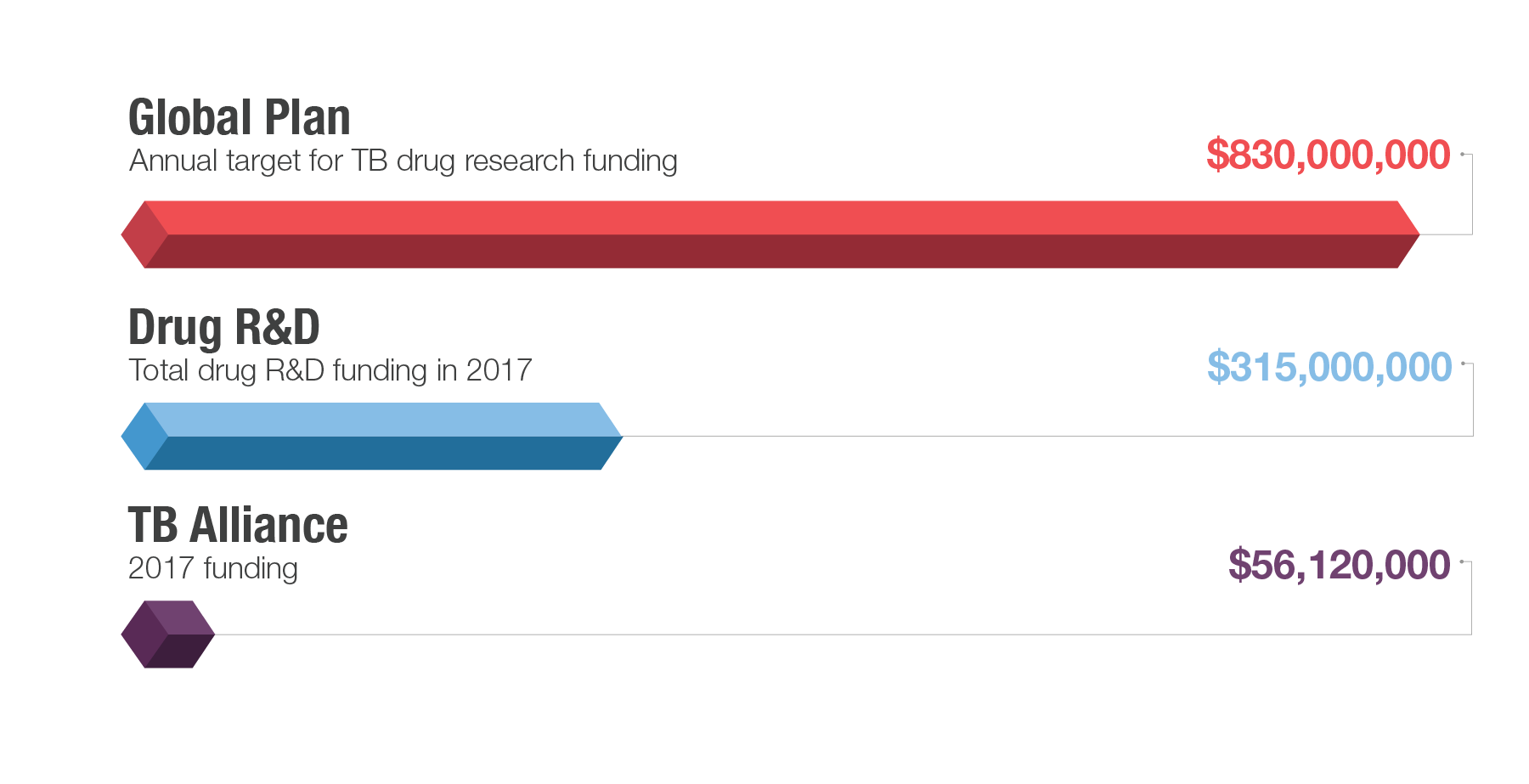
This past year saw new expressions of commitment to invest in TB research and development. Unfortunately, current funding levels remain far short of the $2 billion annual funding target set in the Stop TB Partnership's Global Plan to End TB, 2016–2020, which includes approximately $830 million annually for TB drug research and development.
Treatment Action Group’s 2018 report on TB research funding trends indicated that funding reached $772 million in 2017, with $315 million going to new cures. These figures stand at less than half the overall funding target, both for drugs specifically and research in general. At the United Nations High-Level Meeting on Tuberculosis in 2018, heads of state committed to fill this gap in the meeting’s Political Declaration, though plans to finance this commitment remain outstanding.
Despite a challenging funding environment, TB Alliance has made significant advances, including introducing improved pediatric therapies and submitting its first novel drug for FDA review and approval. It will take a global effort to hold world leaders accountable, but the case is clear: fully funding TB programs would expand and accelerate the development of life-saving treatments for all who suffer from TB.